-
+关注
私聊
-

第11楼2005/04/02
Calibration
A calibration curve is used to determine the unknown
concentration of an element – eg lead – in a solution.
The instrument is calibrated using several
solutions of known concentrations. A calibration
curve is produced which is continually rescaled as
more concentrated solutions are used – the more
concentrated solutions absorb more radiation up to a
certain absorbance. The calibration curve shows the
concentration against the amount of radiation
absorbed (Fig. 8(a)).
The sample solution is fed into the instrument
and the unknown concentration of the element – eg
lead – is then displayed on the calibration curve
(Fig. 8(b)).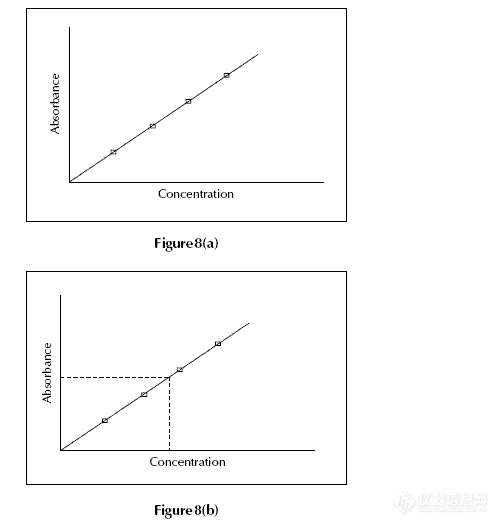
-
+关注
私聊
-

第12楼2005/04/02
Interferences and matrix modification
Other chemicals that are present in the sample may
affect the atomisation process. For example, in flame
atomic absorption, phosphate ions may react with
calcium ions to form calcium pyrophosphate. This
does not dissociate in the flame and therefore results
in a low reading for calcium. This problem is avoided
by adding different reagents to the sample that may
react with the phosphate to give a more volatile
compound that is dissociated easily. Lanthanum
nitrate solution is added to samples containing
calcium to tie up the phosphate and to allow the
calcium to be atomised, making the calcium
absorbance independent of the amount of phosphate.
With electrothermal atomisation, chemical modifiers
can be added which react with an interfering
substance in the sample to make it more volatile than
the analyte compound. This volatile component
vaporises at a relatively low temperature and is
removed during the low and medium temperature
stages of electrothermal atomisation.
-
+关注
私聊
-

第13楼2005/04/02
A bad paint job
Atomic absorption spectrometry is sometimes used for
investigating unusual problems. One such case was
that of a seriously ill baby whose symptoms could not
be explained.
Lead is a toxic element that can cause poisoning
in children. A baby was brought to a hospital
suffering from vomiting and stomach pains, and was
very drowsy. There were no obvious reasons or signs
why the child should be ill.
As part of the routine tests performed, the lead
level in a blood sample from the child was measured
using electrothermal atomisation AAS. The lead
level was higher than normal and there was no
known source for the lead. However, the parents
explained that the child had been chewing the
painted wood on its cot. The paint was also examined
by dissolving it in nitric acid and then using flame
AAS to find out the lead content. A very high level
was found.
Other paints in the baby’s bedroom were found to
have low lead levels. This identified the cot paint as
the source of lead in the baby. The baby’s cot was old
and had been painted when leaded paint was very
common. This type of paint is now banned from
household use and by law all painted toys must be
examined for lead and other toxic metals to make
sure that they are safe for small children.