-
+关注
私聊
-

雾非雾
第13楼2013/02/25
 应助达人
应助达人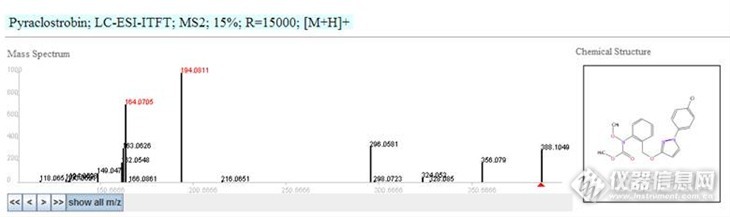 ACCESSION: EA277908RECORD_TITLE: Pyraclostrobin; LC-ESI-ITFT; MS2; 15%; R=15000; [M+H]+DATE: 2012.11.21AUTHORS: Stravs M, Schymanski E, Singer H, Department of Environmental Chemistry, EawagLICENSE: http://massbank.ufz.de/MassBank/files/license.htmlCOPYRIGHT: Copyright (C) 2011 Eawag, Duebendorf, SwitzerlandCOMMENT: CONFIDENCE standard compoundCOMMENT: EAWAG_UCHEM_ID 2779
ACCESSION: EA277908RECORD_TITLE: Pyraclostrobin; LC-ESI-ITFT; MS2; 15%; R=15000; [M+H]+DATE: 2012.11.21AUTHORS: Stravs M, Schymanski E, Singer H, Department of Environmental Chemistry, EawagLICENSE: http://massbank.ufz.de/MassBank/files/license.htmlCOPYRIGHT: Copyright (C) 2011 Eawag, Duebendorf, SwitzerlandCOMMENT: CONFIDENCE standard compoundCOMMENT: EAWAG_UCHEM_ID 2779
CH$NAME: PyraclostrobinCH$NAME: methyl [2-({[1-(4-chlorophenyl)-1H-pyrazol-3-yl]oxy}methyl)phenyl]methoxycarbamateCH$COMPOUND_CLASS: N/A; Environmental StandardCH$FORMULA: C19H18ClN3O4CH$EXACT_MASS: 387.0986CH$SMILES: c1(ccc(n2ccc(OCc3c(cccc3)N(OC)C(OC)=O)n2)cc1)ClCH$IUPAC: InChI=1S/C19H18ClN3O4/c1-25-19(24)23(26-2)17-6-4-3-5-14(17)13-27-18-11-12-22(21-18)16-9-7-15(20)8-10-16/h3-12H,13H2,1-2H3CH$LINK: CAS 175013-18-0CH$LINK: PUBCHEM CID:6422843CH$LINK: INCHIKEY HZRSNVGNWUDEFX-UHFFFAOYSA-NCH$LINK: CHEMSPIDER 4928348
AC$INSTRUMENT: LTQ Orbitrap XL Thermo ScientificAC$INSTRUMENT_TYPE: LC-ESI-ITFTAC$MASS_SPECTROMETRY: MS_TYPE MS2AC$MASS_SPECTROMETRY: IONIZATION ESIAC$MASS_SPECTROMETRY: ION_MODE POSITIVEAC$MASS_SPECTROMETRY: FRAGMENTATION_MODE HCDAC$MASS_SPECTROMETRY: COLLISION_ENERGY 15 % (nominal)AC$MASS_SPECTROMETRY: RESOLUTION 15000AC$CHROMATOGRAPHY: COLUMN_NAME XBridge C18 3.5um, 2.1x50mm, WatersAC$CHROMATOGRAPHY: FLOW_GRADIENT 90/10 at 0 min, 50/50 at 4 min, 5/95 at 17 min, 5/95 at 25 min, 90/10 at 25.1 min, 90/10 at 30 minAC$CHROMATOGRAPHY: FLOW_RATE 200 ul/minAC$CHROMATOGRAPHY: RETENTION_TIME 13.4 minAC$CHROMATOGRAPHY: SOLVENT A water with 0.1% formic acidAC$CHROMATOGRAPHY: SOLVENT B methanol with 0.1% formic acid
MS$FOCUSED_ION: BASE_PEAK 388.1064MS$FOCUSED_ION: PRECURSOR_M/Z 388.1059MS$FOCUSED_ION: PRECURSOR_TYPE [M+H]+MS$DATA_PROCESSING: DEPROFILE SplineMS$DATA_PROCESSING: RECALIBRATE loess on assigned fragments and MS1MS$DATA_PROCESSING: WHOLE RMassBank
PK$ANNOTATION: m/z num {formula mass error(ppm)}118.065 1 C8H8N+ 118.0651 -0.98132.0442 1 C8H6NO+ 132.0444 -1.21133.0521 1 C8H7NO+ 133.0522 -0.79134.0598 1 C8H8NO+ 134.06 -2.02149.047 1 C8H7NO2+ 149.0471 -0.74162.0548 1 C9H8NO2+ 162.055 -0.65163.0626 1 C9H9NO2+ 163.0628 -0.8164.0705 1 C9H10NO2+ 164.0706 -0.46166.0861 1 C9H12NO2+ 166.0863 -1.11194.0811 1 C10H12NO3+ 194.0812 -0.41216.0651 1 C12H10NO3+ 216.0655 -1.71296.0581 1 C19H8N2O2+ 296.058 0.21298.0723 1 C19H10N2O2+ 298.0737 -4.53324.053 1 C17H11ClN3O2+ 324.0534 -1.24328.085 1 C17H15ClN3O2+ 328.0847 0.73356.079 1 C18H15ClN3O3+ 356.0796 -1.9388.1049 1 C19H19ClN3O4+ 388.1059 -2.58PK$NUM_PEAK: 17PK$PEAK: m/z int. rel.int.118.065 8142.3 18132.0442 12531.4 28133.0521 6314.9 14134.0598 16177.1 36149.047 39426.3 88162.0548 81735.3 182163.0626 141230.8 315164.0705 317833.8 710166.0861 5274.5 11194.0811 446803.4 999216.0651 3872.5 8296.0581 146287 327298.0723 2417 5324.053 19974.7 44328.085 3734.9 8356.079 81956.1 183388.1049 133076.1 297//