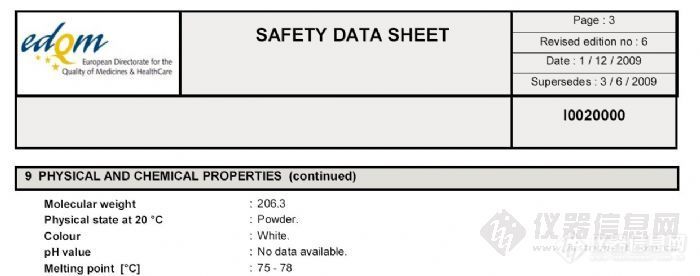
-
+关注
私聊
-

poorlittle
第12楼2011/10/09
From NIST :
……The terms atomic weight and molecular weight are obsolete and thus should be avoided. They have been replaced by the equivalent but preferred terms relative atomic mass, symbol Ar, and relative molecular mass, symbol Mr, respectively [4: ISO 31-8]……
(see 8.4 at http://physics.nist.gov/Pubs/SP811/sec08.html )
However, the term “molecular weight” is still used in NIST webbook (see http://webbook.nist.gov/chemistry/mw-ser.html ). I think that “relative molecular mass” may be more scientific, but the traditionally used term “molecular weight” is still acceptable.
-
+关注
私聊
-

poorlittle
第15楼2011/10/12
It was over 40 years ago when I learnt the term “Molecular Weight” in high school. During that time, we also frequently used公斤 instead of千克.
Would 欧盟的法规 change later?poorlittle(poorlittle) 发表:I am a layman in chemistry. However, if I have to choose between “mass” and “weight”, I prefer “mass” because “weight” involves the gravitational acceleration in general physics (W=m×g).
But when I was in high school, it was called “Molecular Weight”
It is said “为了避免质量和重量的混淆,所以,国际计量大会建议在科学用语中,取消重量概念。” (please refer to 28#, http://bbs.instrument.com.cn/shtml/20110712/3410991/index_3.shtml )
-
+关注
私聊
-

rock_rock
第16楼2011/10/14
All the three terms are different:
molar mass = 摩尔质量 = the mass of 1 mole substances (unit = kg/mol or g/mol)
molecular weight = 相对分子质量 = the ratio of the mass of the molecule to 1/12 of the mass of carbon 12.Since it is a ratio, it has no unit.
molecular mass =分子量/分子质量 = the mass of a single molecule/atom (unit = amu or Da, 1amu = 1/12 mass of a C-12 atom)
However, some of the terms are already outdated. Molecular weight is a ratio instead of "weight". It has been replaced by "relative molecular mass".
In Chinese, 分子量 was actually used to represent "molecular weight", therefore, the terms has been regulated as 相对分子质量.
There are more complicated stories behind molecular mass. Therefore, contemporary science is avoiding to use this term any more.
Actually, the value of molecular weight = molecular mass. The differences are just on the definitions and the units.
-
+关注
私聊
-

rock_rock
第17楼2011/10/14
40 years ago, SI units were not introduced yet. After the adoption of SI units, the use of kilo-, mega-, giga-, etc. has been standardized.
poorlittle(poorlittle) 发表:It was over 40 years ago when I learnt the term “Molecular Weight” in high school. During that time, we also frequently used公斤 instead of千克.
Would 欧盟的法规 change later?