
方案摘要
方案下载| 应用领域 | 能源/新能源 |
| 检测样本 | 锂电池 |
| 检测项目 | |
| 参考标准 | EQCM-D |
本文作者开发了一种高度敏感的基于EQCM-D方法的,在三种不同的锂电池电解质溶液中,原位测量了由于锂离子插入/提出以及固体电解质界面的形成和生长引起的复合LTO电极的重量和粘弹性的变化。利用声学多层形式主义建立一个自洽的,复合LTO电极的粘弹性模型,描述了在气体空气环境下,与锂电池电解质溶液在开路电压下以及外加电压下相接触条件下共振率和共振宽度的变化。该模型选择性地表征了多层复合电极组件中的每一层(电极的刚性和粘弹性部分+固体电解质界面)与电解质溶液接触时的机械状态。EQCM-D方法可实现实时无损探测电极粘弹性,厚度剪切及谐振模式。通过一种先进的粘弹性模型来拟合EQCM-D的实验数据,可以得到每层的重量和粘弹性参数。我们的主要发现是,锂电池电解液及添加剂可通过EQCM-D短时间实验进行筛选。同时,通过探测电极容量保持率,量化了固体电解质界面膜生长的本征粘弹性特性。我们的结论是,使用适量的水淹没研究EQCM-D腔体,可能提供有关实际电极的循环性能的信息的重要预测信息,在不同电解液中对纽扣电池进行测试时。所开发的实验装置和建模程序可应用于各种类型的离子插入电极,固体电解质界面型保护膜,包括低压阳极和高压阴极。我们目前正在致力于改进EQCM-D方法将Sflood(溶液质量与浸入电极质量之比)减小一个数量级,这足够为EQCM-D技术广泛应用于先进储能研究领域开启一扇新的大门。
本文作者开发了一种高度敏感的基于EQCM-D方法的,在三种不同的锂电池电解质溶液中,原位测量了由于锂离子插入/提出以及固体电解质界面的形成和生长引起的复合LTO电极的重量和粘弹性的变化。利用声学多层形式主义建立一个自洽的,复合LTO电极的粘弹性模型,描述了在气体空气环境下,与锂电池电解质溶液在开路电压下以及外加电压下相接触条件下共振率和共振宽度的变化。该模型选择性地表征了多层复合电极组件中的每一层(电极的刚性和粘弹性部分+固体电解质界面)与电解质溶液接触时的机械状态。EQCM-D方法可实现实时无损探测电极粘弹性,厚度剪切及谐振模式。通过一种先进的粘弹性模型来拟合EQCM-D的实验数据,可以得到每层的重量和粘弹性参数。我们的主要发现是,锂电池电解液及添加剂可通过EQCM-D短时间实验进行筛选。同时,通过探测电极容量保持率,量化了固体电解质界面膜生长的本征粘弹性特性。我们的结论是,使用适量的水淹没研究EQCM-D腔体,可能提供有关实际电极的循环性能的信息的重要预测信息,在不同电解液中对纽扣电池进行测试时。所开发的实验装置和建模程序可应用于各种类型的离子插入电极,固体电解质界面型保护膜,包括低压阳极和高压阴极。我们目前正在致力于改进EQCM-D方法将Sflood(溶液质量与浸入电极质量之比)减小一个数量级,这足够为EQCM-D技术广泛应用于先进储能研究领域开启一扇新的大门。
Authors:Vadim Dargel, NetanelShpigel, Sergey Sigalov, Prasant Nayak, Mikhael D. Levi ,LeonidDaikhin and Doron Aurbach
Journal:Nature Communications
DOI: 10.1038/s41467-017-01722-x
Abstract:锂离子电池电极表面形成的固体-电解质界面在控制其循环性能方面起着关键作用。虽然各种表面敏感的光谱和显微镜技术用来对其特性进行表征,重点关注的是表面物性而不是表面薄膜的力学性能。在这里,我们报道一种高度敏感的探测复合材料Li4Ti5O12电极与锂离子嵌入电极表面形成薄膜的重量和粘弹性的方法。耗散型电化学石英晶体微天平用来监测并测量LiTFSI, LiPF6,and LiPF6?+?2%碳酸亚乙烯酯溶液,通过对多层粘弹性模型的拟合,得到表面膜的结构参数。只有几个快速的周期需要限定表面薄膜在Li4Ti5O12阳极,并按着顺序升高LiPF6?<?LiPF6?+?2%vinylene carbonate << LiTFSI。
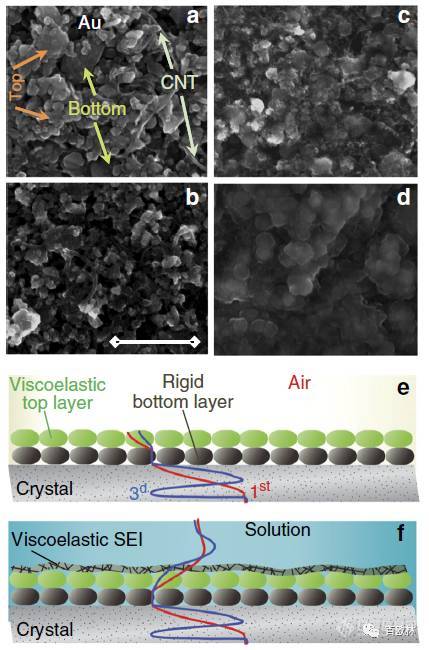
Fig. 1 SEM images of LTO electrodes on quartz-crystal surface and their model presentation. a Pristine electrode, b cycled in EC-DMC 1M LiTFSI, c 1M LiPF6, and d 1M LiPF6 + 2% VC solutions (scale bar = 500 nm). e Sketch of velocity profiles for rigid and viscoelastic layers of LTO electrodes in air and f in electrolyte solution. Velocity profile of shear wave crossing the multilayer assembly is exemplified for fundamental frequency and third overtone order
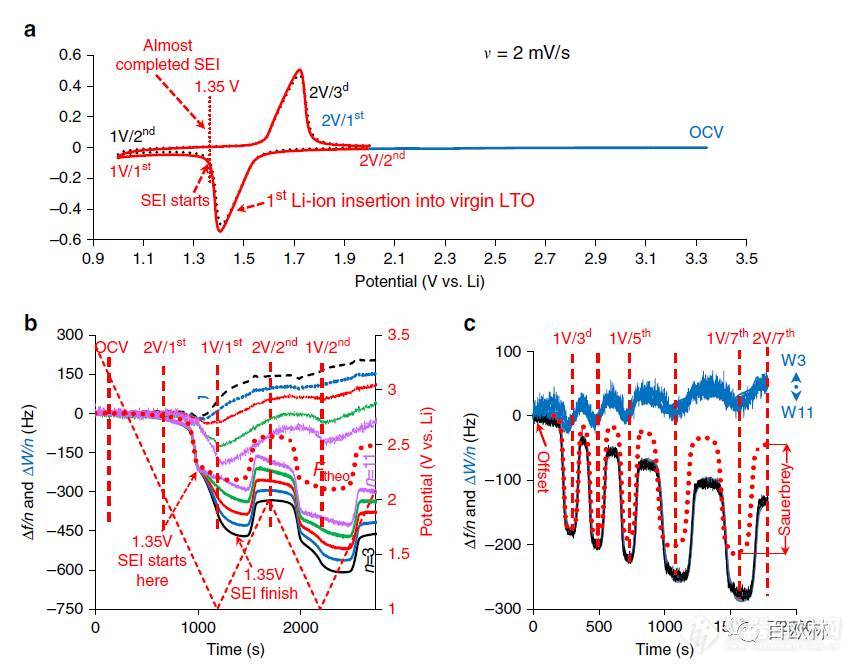
Fig. 2 Characterization of LTO electrodes in EC-DMC 1M LiTFSI solutions. a Two first sequential CV cycles from OCV to 1 V and then to 2 V, as indicated. The formation of SEI starts at 1.35 V during the first discharge and is almost completed at 1.35 V during the subsequent charge. b, c Frequency and resonance width changes at different overtones, n (shown by different colors) as functions of time during the first two initial cycles and during the subsequent five cycles, respectively. The dotted red line (Ftheo) denotes the frequency shift calculated from the intercalation charge using the Faraday law and the Sauerbrey equation. The dashed red lines denote the potentials at which viscoelastic analysis has been carried out. In b W3?W11 denotes the range of the resonance width changes for odd harmonics from n = 3 to 11
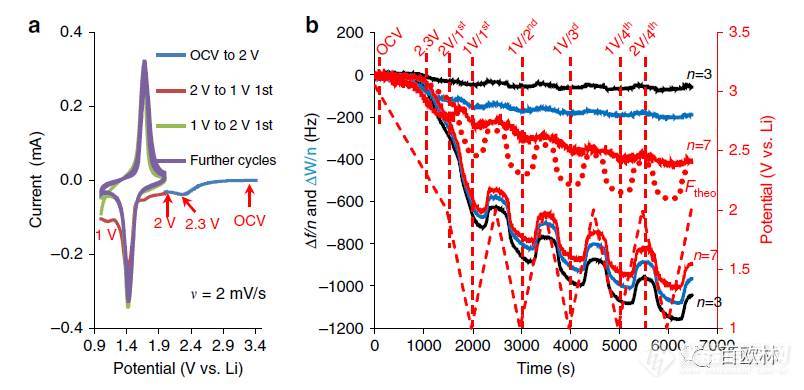
Fig. 3 Characterization of LTO electrodes in EC-DMC 1M LiPF6 solutions. a Two first sequential CVs from OCV to 1 V and then to 2 V, as indicated. b Frequency and resonance width changes at different overtones (shown by different colors) as functions of time during electrode cycling. The dashed red lines denote the potentials at which viscoelastic analysis has been carried out
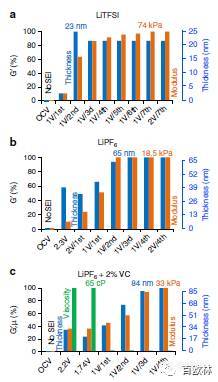
Fig. 4 Viscoelastic parameters of LTO electrodes when in contact with different electrolyte solutions: a 1M LiTFSI, b 1M LiPF6, and c 1M LiPF6 +2% VC. Storage modulus and thickness of the SEI on LTO electrode at different potentials are shown by orange and blue columns, respectively. The VC green columns show the dynamic viscosity of SEI. The full set of parameters for all three systems is listed in Supplementary Tables 1, 2 and 3, respectively
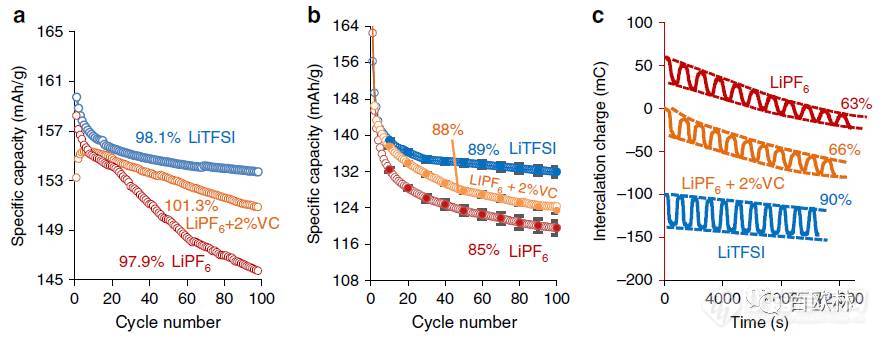
Fig. 5 Cycling experiments of LTO electrodes in cells with different floodness factor, Sflood. Cycling in coin cells at 1C rate a, b and in experimental EQCM-D cell (scan rate 2 mV/s) in three electrolyte solutions as indicated. Capacity retention for LTO electrodes in all three solutions are referred to 10th cycle and expressed as % with respect to the capacity during the first charge. The ratio of LTO mass to the mass of the solutions were equal to: a 5mg LTO/50 mg solution (Sflood = 10); b 80 μg LTO/50 mg solution (Sflood = 6.25 × 102); c 90 μg LTO/1 g solution (Sflood = 1.10 × 104). For clarity, in c the intercalation charge is shifted by 60 and ?100 mC for LiPF6 and LiTFSI solutions, respectively

QSense耗散型石英晶体微天平在制药领域中的应用
使用QCM-D 进行清洁研究
海南大学化工学院李嘉诚教授利用石英晶体微天平技术研究光响应性海藻酸基大分子表面活性剂与疏水叶片表面的相互作用
相关产品
Attension Theta Flow 光学接触角测量仪
QSense High Pressure 高压石英晶体微天平
QSense全自动八通道石英晶体微天平
QSense卓越版四通道石英晶体微天平
QSense Explorer扩展版石英晶体微天平
KSV NIMA roll to roll 柔性LB膜制备系统
KSV NIMA LB膜分析仪
Attension Theta Flex 光学接触角仪
KSV NIMA Microbam 独立式小型布鲁斯特角显微镜
KSV NIMA 布鲁斯特显微镜
KSV NIMA常规 交替型LB膜分析仪
KSV NIMA 缎带型Langmuir膜分析仪
KSV NIMA Langmuir膜分析仪
QSense 石英晶体微天平定制表面芯片
高温高压表面/界面测量系统

关注
拨打电话

留言咨询