
 400-659-9826
400-659-9826
 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询

 400-860-5168转1594
400-860-5168转1594
 留言咨询
留言咨询

 400-860-5168转3128
400-860-5168转3128
 留言咨询
留言咨询

 400-860-5168转3450
400-860-5168转3450
 留言咨询
留言咨询

 400-860-5168转4648
400-860-5168转4648
 留言咨询
留言咨询

 400-860-5168转1270
400-860-5168转1270
 留言咨询
留言咨询

 400-860-5168转4406
400-860-5168转4406
 留言咨询
留言咨询

 留言咨询
留言咨询

 400-860-5168转3662
400-860-5168转3662
 留言咨询
留言咨询

 400-860-5168转2882
400-860-5168转2882
 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询

 400-860-5168转6121
400-860-5168转6121
 留言咨询
留言咨询

 400-860-5168转1200
400-860-5168转1200
 留言咨询
留言咨询

 400-860-5168转4406
400-860-5168转4406
 留言咨询
留言咨询

 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询
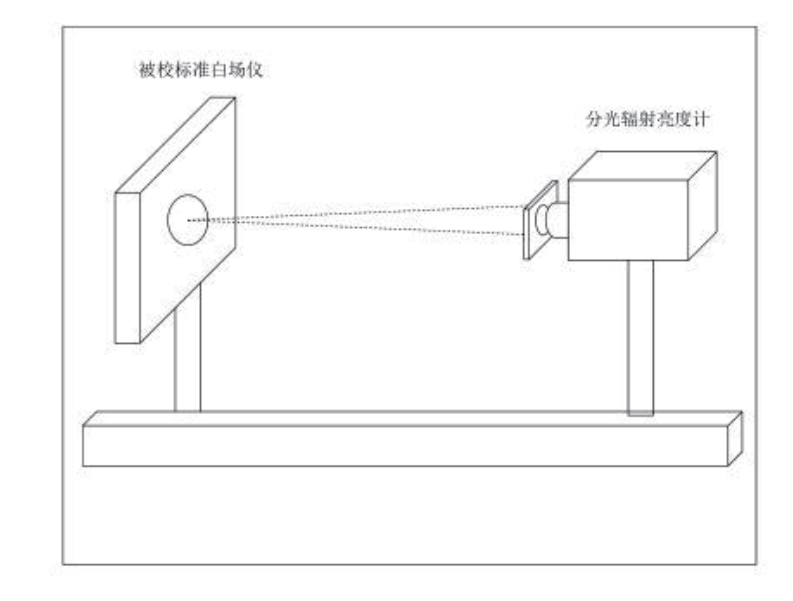
 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询

 400-860-5168转2943
400-860-5168转2943
 留言咨询
留言咨询

 400-860-5168转4648
400-860-5168转4648
 留言咨询
留言咨询

 留言咨询
留言咨询

 400-860-5168转3662
400-860-5168转3662
 留言咨询
留言咨询

 400-860-5168转0843
400-860-5168转0843
 留言咨询
留言咨询

 400-812-0970
400-812-0970
 留言咨询
留言咨询

 400-860-5168转2882
400-860-5168转2882
 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询
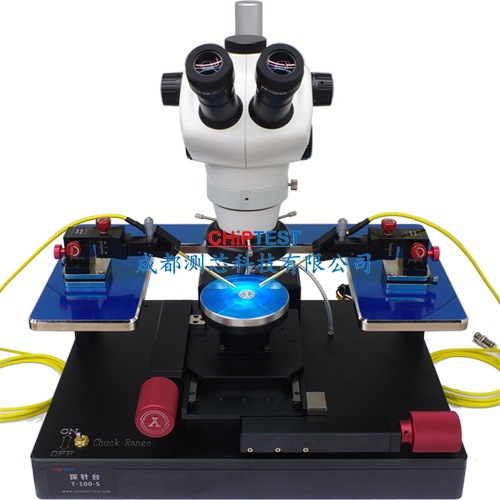
 留言咨询
留言咨询

 400-860-5168转1887
400-860-5168转1887
 留言咨询
留言咨询

 400-860-5168转4642
400-860-5168转4642
 留言咨询
留言咨询