方案详情文
智能文字提取功能测试中
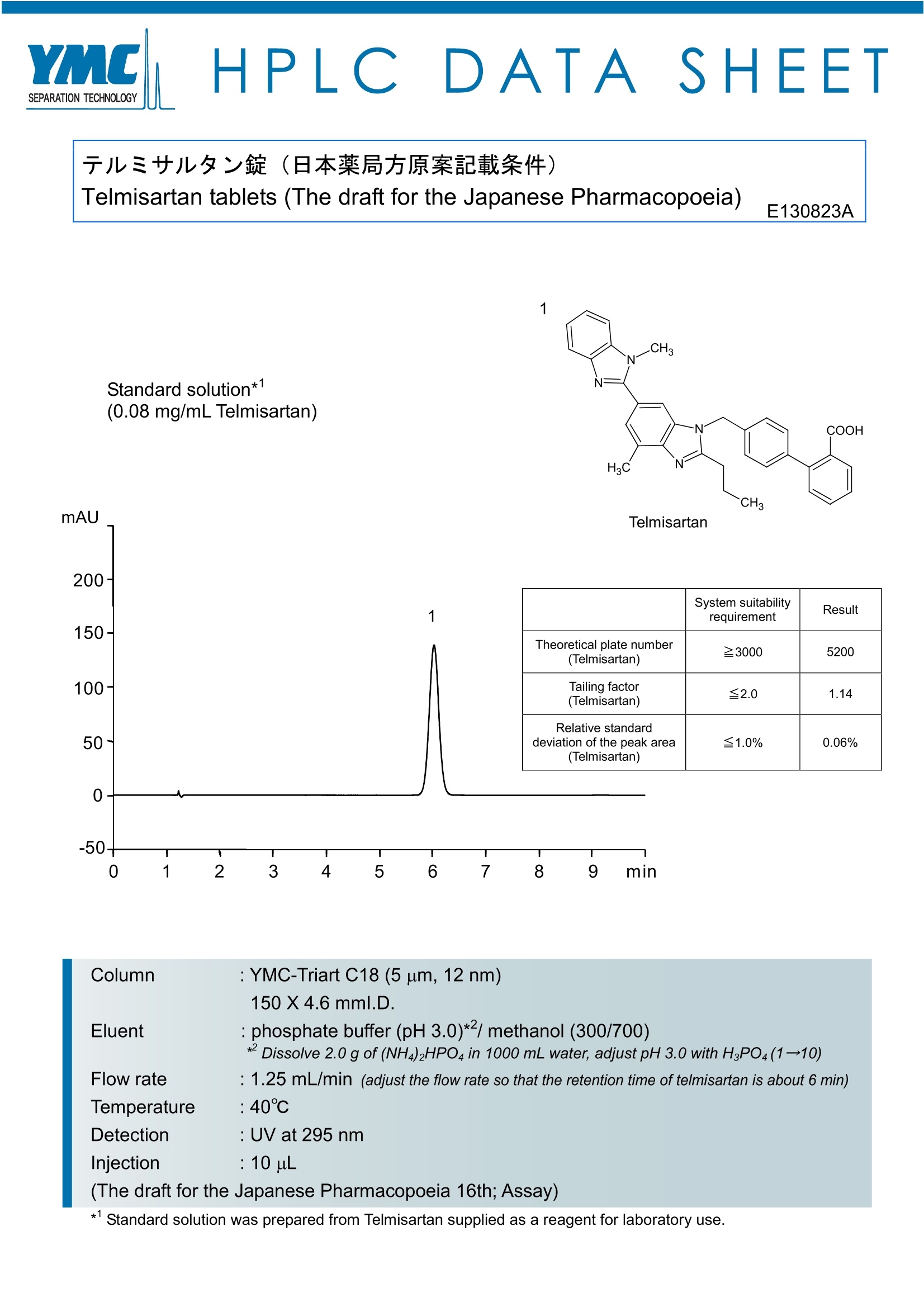
HPLCDATA:SHEET 于儿三廿儿夕沙锭(日本藥局方原案記載条件) Telmisartan tablets (The draft for the Japanese Pharmacopoeia) E130823A Standard solution*(0.08 mg/mL Telmisartan) mAU Telmisartan 200 1 150- 100- 50) System suitabilityrequirement Result Theoretical platenumber(Telmisartan) 23000 5200 Tailing factor(Telmisartan) ≤2.0 1.14 Relative standarddeviation of the peak area(Telmisartan) ≤1.0% 0.06% 0 -50- 0 1 2 3 4 5 6 7 8 9 min Column : YMC-Triart C18 (5 um, 12nm) 150X4.6mml.D. Eluent : phosphate buffer (pH 3.0)*2/ methanol (300/700) *Dissolve 2.0 g of (NH4)2HPO4in 1000 mL water, adjust pH 3.0 with H3P04(1→10) Flow rate : 1.25 mL/min (adjust the flow rate so that the retention time of telmisartan is about 6 min)Temperature : 40℃ Detection : UV at 295 nm Injection : 10 uL (The draft for the Japanese Pharmacopoeia 16th;Assay) *1Standard solution was prepared from Telmisartan supplied as a reagent for laboratory use.
关闭-
1/1
产品配置单
深圳凯米斯科技有限公司为您提供《替米沙坦片中含量检测方案(液相色谱柱)》,该方案主要用于化药制剂中含量测定检测,参考标准《暂无》,《替米沙坦片中含量检测方案(液相色谱柱)》用到的仪器有null。
我要纠错
相关方案


 咨询
咨询





