

暴走追风 2009/03/03
[quote]原文由 [B]wxlsfl[/B] 发表: [quote]原文由 [B]zhk521[/B] 发表: [img]https://www.instrument.com.cn/bbs/images/affix.gif[/img][url=http://www.instrument.com.cn/bbs/download.asp?ID=136424]HPLC法测定阿司匹林中游离水杨酸含量[/url][/quote] 坤哥,文件我下不来!![/quote] 换个电脑 我这可慢了
jun来也 2009/03/15
阿司匹林(乙酰水杨酸)的合成(实验室) [img]https://www.instrument.com.cn/bbs/images/affix.gif[/img][url=http://www.instrument.com.cn/bbs/download.asp?ID=138606]阿司匹林(乙酰水杨酸)的合成[/url]
〓疯子哥〓 2009/03/03
一般工艺流程可能不会向别人透露的~都需要花高价钱购买或者别人转让的~
jonye886 2009/03/03
[img]https://ng1.17img.cn/bbsfiles/images/2009/03/200903031553_136434_1616412_3.jpg[/img]
遨翔的岁月 2009/03/10
[quote]原文由 [B]wxlsfl[/B] 发表: [quote]原文由 [B]zhk521[/B] 发表: [img]https://www.instrument.com.cn/bbs/images/affix.gif[/img][url=http://www.instrument.com.cn/bbs/download.asp?ID=136424]HPLC法测定阿司匹林中游离水杨酸含量[/url][/quote] 坤哥,文件我下不来!![/quote] 资料中心有游离水杨酸含量的测定方法。
拉拉与玻璃
第3楼2009/03/03
坤哥,文件我下不来!!
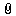 HPLC法测定阿司匹林中游离水杨酸含量
HPLC法测定阿司匹林中游离水杨酸含量lingzhong
第9楼2009/03/11
Assay for butalbital and aspirin and limit of free salicylic acid—
Mobile phase—Prepare a suitable filtered and degassed mixture of water, acetonitrile, and phosphoric acid (3100:725:4). Adjust the ratio as necessary.
Solvent mixture—Mix 40 mL of formic acid and 4000 mL of acetonitrile.
Butalbital standard stock solution—Dissolve an accurately weighed quantity of USP Butalbital RS in Solvent mixture to obtain a solution having a known concentration of about 3250Jµg per mL, J being the ratio of the labeled amount, in mg, of butalbital to the labeled amount, in mg, of aspirin per tablet.
Salicylic acid standard stock solution—Dissolve an accurately weighed quantity of USP Salicylic Acid RS in Solvent mixture to obtain a solution having a known concentration of about 200 µg per mL.
Butalbital and salicylic acid standard preparation—Transfer 25.0 mL of Butalbital standard stock solution and 3.0 mL of Salicylic acid standard stock solution to a 250-mL volumetric flask, dilute with Solvent mixture to volume, and mix. This solution contains about 325Jµg of butalbital and 2.4 µg of salicylic acid per mL.
Aspirin standard preparation—Dissolve an accurately weighed quantity of USP Aspirin RS in Solvent mixture to obtain a solution having a known concentration of about 325 µg per mL.
Resolution solution—Transfer 4.0 mL of Butalbital standard stock solution and 3.0 mL of Salicylic acid standard stock solution to a 50-mL volumetric flask, dilute with Solvent mixture to volume, and mix.
Assay preparation—Weigh and finely powder not fewer than 20 Tablets. Transfer an accurately weighed portion of the powder, equivalent to about 80 mg of aspirin, to a 250-mL volumetric flask, dilute with Solvent mixture to volume, sonicate for 15 minutes, and mix. Pass a portion of this solution through a 0.5-µm porosity filter before use.
Chromatographic system (see Chromatography 621 )—The liquid chromatograph is equipped with a 214-nm detector and a 3.9-mm × 30-cm column that contains 10-µm packing L1. The flow rate is about 1.5 mL per minute. Chromatograph the Butalbital and salicylic acid standard preparation, the Aspirin standard preparation, and the Resolution solution as directed for Procedure: the resolution, R, between the butalbital and salicylic acid peaks is not less than 3.0; and the relative standard deviation for replicate injections of the Standard preparations is not more than 3.0% for butalbital and aspirin, and not more than 6.0% for salicylic acid.
Procedure—Separately inject equal volumes (about 10 µL) of the Standard preparations and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks, and for the minor peak corresponding to salicylic acid. The relative retention times are about 0.6 for aspirin, 0.85 for salicylic acid, and 1.0 for butalbital. [NOTE—After use, the column may be regenerated by passing through it at least 50 mL of a mixture of acetonitrile, methanol, and water (1:1:1), followed by a mixture of acetonitrile and water (1:1).] Calculate the quantity, in mg, of butalbital (C11H16N2O3) in the portion of Tablets taken by the formula:
0.25C(rU / rS)
in which C is the concentration, in µg per mL, of USP Butalbital RS in the Butalbital and salicylic acid standard preparation; and rU and rS are the butalbital peak responses obtained from the Assay preparation and the Butalbital and salicylic acid standard preparation, respectively. Calculate the quantity, in mg, of aspirin (C9H8O4) in the portion of Tablets taken by the formula:
0.25C(rU / rS)
in which C is the concentration, in µg per mL, of USP Aspirin RS in the Aspirin standard preparation; and rU and rS are the aspirin peak responses obtained from the Assay preparation and the Aspirin standard preparation, respectively. Calculate the percentage of free salicylic acid in the Tablets taken by the formula:
25(C / a)(rU / rS)
in which C is the concentration, in µg per mL, of the USP Salicylic Acid RS in the Butalbital and salicylic acid standard preparation; a is the quantity, in mg, of aspirin in the portion of Tablets taken, based on the labeled amount; and rU and rS are the salicylic acid peak responses obtained from the Assay preparation and the Butalbital and salicylic acid standard preparation, respectively: not more than 3.0% is found.
lingzhong
第10楼2009/03/11
Limit of free salicylic acid—[NOTE—Use glassware in this test.]
Solvent—Add 1 mL of phosphoric acid to 1000 mL of methanol, and mix.
Standard preparation—Prepare a solution of USP Salicylic Acid RS in Solvent having a known concentration of about 0.0012 mg per mL. Use this solution promptly.
Test preparation—[NOTE—Perform this test on the same day the tablets are powdered.] Weigh and finely powder not fewer than 20 Tablets. Transfer an accurately weighed portion of the powder, equivalent to about 65 mg of aspirin, to a 200-mL flask, add 100.0 mL of Solvent, and shake by mechanical means for 15 minutes. Filter a portion of this solution, discarding the first 15 mL of the filtrate, and use the clear filtrate as the Test preparation. Use this solution within 20 minutes after the addition of the Solvent.
Procedure—Immediately place the cell containing the solution under test in the cell compartment of a spectrophotofluorimeter, and allow to equilibrate for 2 minutes. Concomitantly measure the intensities of the fluorescence of the Test preparation and the Standard preparation at 444 nm, using an excitation wavelength of 305 nm. Calculate the percentage of salicylic acid in the portion of Tablets taken by the formula:
10,000(C / a)(IU / IS)
in which C is the concentration, in mg per mL, of USP Salicylic Acid RS in the Standard preparation; a is the quantity, in mg, of aspirin in the portion of Tablets taken to prepare the Test preparation, based on the labeled amount; and IU and IS are the fluorescence intensity readings obtained from the Test preparation and the Standard preparation, respectively. [NOTE—If the intensity of the Test preparation greatly exceeds that of the Standard preparation, immediately transfer 5.0 mL of the Test preparation to a 50-mL volumetric flask, dilute with Solvent to volume, and mix. Immediately determine the intensity of this solution, and calculate the percentage of salicylic acid in the portion of Tablets taken by the formula: 100,000(C/a)(IU / IS).] Not more than 3.0% is found.