方案详情文
智能文字提取功能测试中
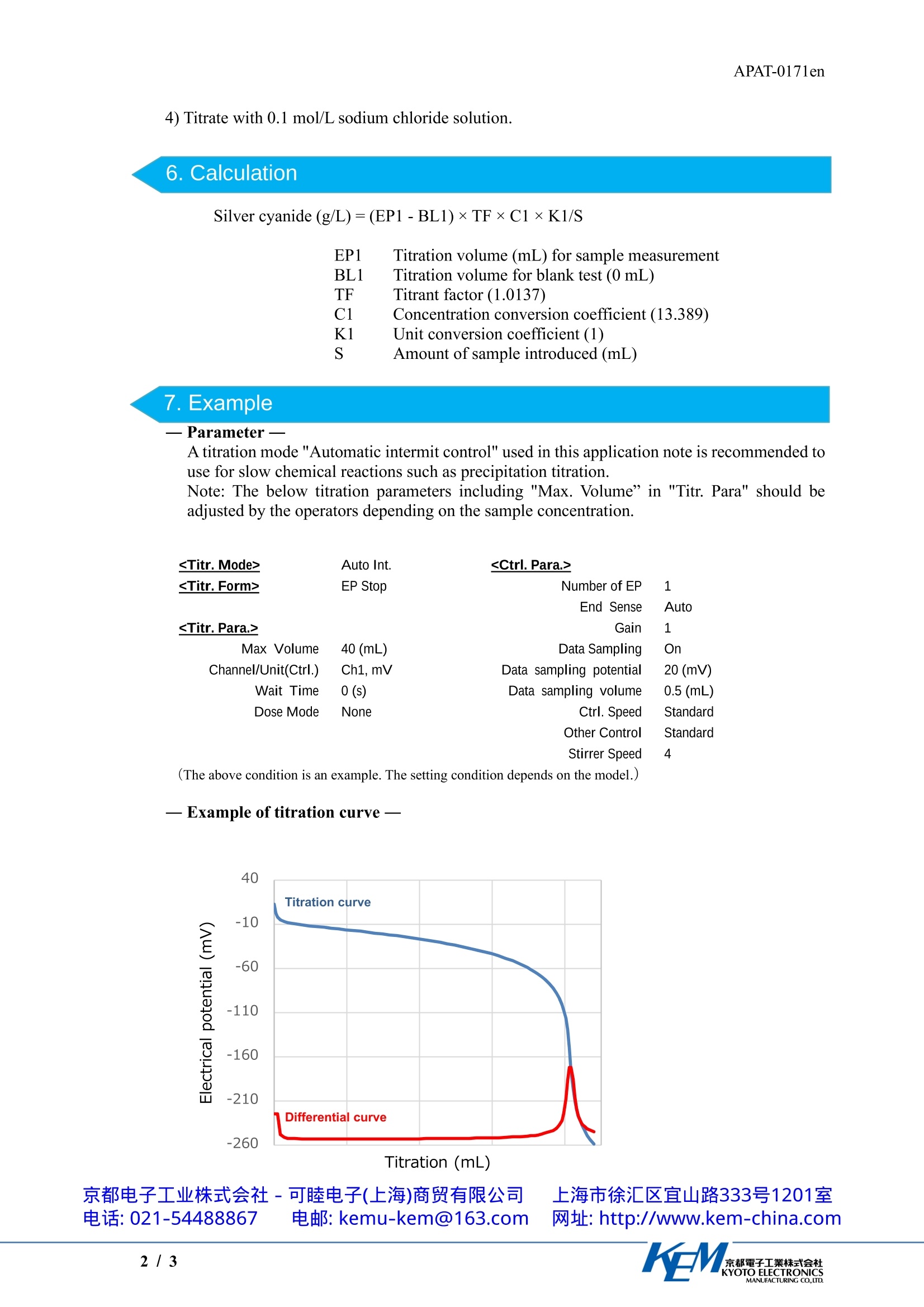
APAT-0171en京都電子工業株式会社KYOTO ELECTRONICSMANUFACTURING CO.,LTD. 4) Titrate with 0.1 mol/L sodium chloride solution. Application Note Determination of silver cyanide in silver platingsolution Industry MetalsInstrument Automatic potentiometric titratorMeasurement method Potentiometric titration / Precipitation titration Standards 1.Scope Warning ·Toxic gas and Exothermic reaction The decomposition of cyanide ion by sulfuric acid and nitric acid releases hydrogencyanide via an exothermic reaction. Decomposition operations should be performedin fume hoods, and protective masks and gloves should be worn. The concentration of silver cyanide in the plating bath greatly affects the quality of silver plating.Therefore, controlling its concentration is extremely important. The concentration of silvercyanide can be determined by decomposing the cyanide ions and titrating the silver ions producedwith a sodium chloride standard solution (Note 1). Ag++CI→AgCI This application shows an example of the determination of silver cyanide in a silver platingsolution. 2. Post-measurement procedure ·Polish the detection area of the combined silver electrode with polishing paper or other material.·Reference internal liquid (the combined silver electrode) is replaced about once every two weeks. 3.Apparatus Main unit Automatic potentiometric titrator (Preamplifier STD)Electrode Combined silver electrode (Reference internal solution1mol/Lpotassium nitrate solution) 4.Reagents Titrant 0.1mol/L Sodium chloride solutionAdditive reagents Concentrated sulfuric acid, Concentrated nitric acid 5 Procedure 1) Add exactly 10 mL of the sample into a beaker. 2) Warning (Hydrogen cyanide and Exothermic reaction) Add 20 mL of concentrated sulfuric acid and three drops of concentrated nitric acid and thengently stir to induce the reaction. 3) After the solution cools to room temperature, add 100 mL of pure water and stir until allsolids dissolve. 6.Calculation Silver cyanide (g/L)=(EP1-BL1)×TF×C1×K1/S EP1 Titration volume (mL) for sample measurement BL1 Titration volume for blank test (0 mL) F Titrant factor (1.0137) Concentration conversion coefficient (13.389) Unit conversion coefficient (1) S Amount of sample introduced (mL) 7. Example 一Parameter一 A titration mode "Automatic intermit control" used in this application note is recommended touse for slow chemical reactions such as precipitation titration. Note: The below titration parameters including "Max. Volume” in "Titr. Para" should beadjusted by the operators depending on the sample concentration.
-
1/3

-
2/3
还剩1页未读,是否继续阅读?
继续免费阅读全文产品配置单
可睦电子(上海)商贸有限公司-日本京都电子(KEM)为您提供《镀银溶液中氰化银检测方案(自动电位滴定)》,该方案主要用于其他中其他检测,参考标准《暂无》,《镀银溶液中氰化银检测方案(自动电位滴定)》用到的仪器有AT-710M四通道旗舰型自动电位滴定仪、AT-710S豪华型自动电位滴定仪。
我要纠错
推荐专场
相关方案






 咨询
咨询






