

Onway
第3楼2010/03/10
第四阶段 原子吸收分析仪器的发展
随着原子吸收技术的发展,推动了原子吸收仪器的不断更新和发展,而其它科学技术进步,为原子吸收仪器的不断更新和发展提供了技术和物质基础。近年来,使用连续光源和中阶梯光栅,结合使用光导摄象管、二极管阵列多元素分析检测器,设计出了微机控制的原子吸收分光光度计,为解决多元素同时测定开辟了新的前景。微机控制的原子吸收光谱系统简化了仪器结构,提高了仪器的自动化程度,改善了测定准确度,使原子吸收光谱法的面貌发生了重大的变化。联用技术(色谱-原子吸收联用、流动注射-原子吸收联用)日益受到人们的重视。色谱-原子吸收联用,不仅在解决元素的化学形态分析方面,而且在测定有机化合物的复杂混合物方面,都有着重要的用途,是一个很有前途的发展方向。
Onway
第4楼2010/03/10
另外
原子吸收光谱法的优点与不足
<1> 检出限低,灵敏度高。火焰原子吸收法的检出限可达到ppb级,石墨炉原子吸
收法的检出限可达到10-10-10-14g。
<2> 分析精度好。火焰原子吸收法测定中等和高含量元素的相对标准差可<1%,其准
确度已接近于经典化学方法。石墨炉原子吸收法的分析精度一般约为3-5%。
<3> 分析速度快。原子吸收光谱仪在35分钟内,能连续测定50个试样中的6种元素。
<4> 应用范围广。可测定的元素达70多个,不仅可以测定金属元素,也可以用间接
原子吸收法测定非金属元素和有机化合物。
<5> 仪器比较简单,操作方便。
<6> 原子吸收光谱法的不足之处是多元素同时测定尚有困难,有相当一些元素的测
定灵敏度还不能令人满意
Onway
第5楼2010/03/10
连续光源原子吸收光谱仪发展历史(英文)
Historical Development of Continuum Source Atomic Absorption Spectrometry
When Bunsen and Krichhoff were carrying out their systematic investigation of the ‘line reversal’ in alkali and alkaline earth elements, i.e. the correlation between emission and absorption of radiation by atoms, in the early 1860s, they used a continuum source, i.e. ‘white light’, for their absorption measurements. The few researchers that used atomic absorption for their investigations in the second half of the 19th century, such as Lockyer, used similar equipment, as shown in Figure 1.1, for obvious reasons: Firstly, continuum light sources were the only reliable sources available at that time, and secondly, they served perfectly the purpose of detecting and measuring the ‘black lines’, i.e. the interruptions in the otherwise continuous spectrum, caused by atomic absorption.
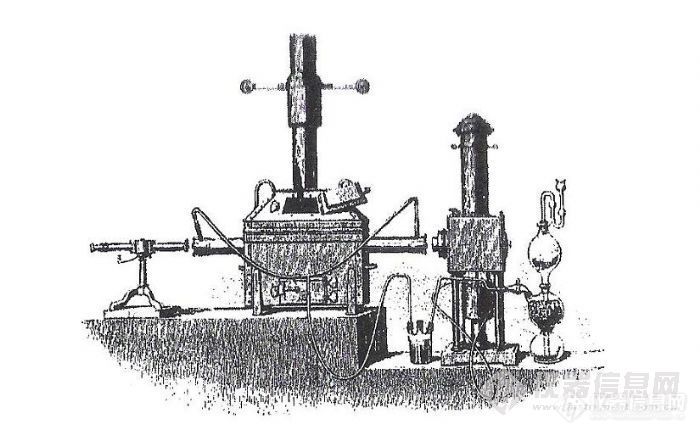
Figure 1.1: Apparatus used by Lockyer for atomic absorption measurements: light source on the right; atomizer in the middle (iron tube mounted in a coal-fired furnace, while hydrogen was generated in a Kipp’s apparatus to provide a reducing atmosphere); spectroscope on the left
Onway
第6楼2010/03/10
In the first half of the 20th century, when atomic spectra were increasingly used not only for the qualitative identification, but also for the quantitative determination of elements, it was at least in part because of this continuum source that spectroscopists gave preference to atomic emission over atomic absorption. It is obviously much easier to detect a small radiation in front of a non-emitting, ‘black’ background, than a small reduction the detector, as was common practice at that time, it is much easier to quantify a small increase in the opacity (‘blackening’) of the photographic layer than a small decrease in the opacity of an otherwise black plate. Hence, the radiation source was obviously the reason why atomic absorption was essentially excluded from analytical atomic spectroscopy for more than half a century.
It was only in 1952 when Alan Walsh, after having worked on the spectrochemical analysis of metals for seven years, and in molecular spectroscopy for another six years, began to wonder why molecular spectra were usually obtained in absorption and atomic spectra in emission. The conclusion of his musing was that there was no good reason for neglecting atomic absorption spectra. Obviously, Walsh also had to consider the question of the proper radiation source for recording atomic absorption spectra, and he came to the conclusion that a resolution of approximately 2 pm would be required if a continuum source was used. This was far beyond the capabilities of the best spectrometer available in his laboratory at the time, and he concluded that ‘One of the main difficulties is due to the fact that the relations between absorption and concentration depend on measurement of atomic absorption requires line radiation sources with the sharpest possible emission lines. The task of monochromator is then merely to separate the line used for the measurement from all the other lines emitted by the source. The high-resolution demand for atomic absorption measurements is thus provide by the line source.
Onway
第7楼2010/03/10
Anyway, Walsh was quite fortunate because, although the hollow cathode glow had already been discovered back in 1916 by Paschen, and had since been used as a fine-line source for spectroscopic investigations, it was only in 1955 that the first sealed-off hollow cathode lamp was constructed. Without this development and the significant amount of research that Walsh and colleagues put into the improvement of hollow cathode lamp design, atomic absorption spectrometry (AAS) would probably not have been accepted as a routine technique to the same extent, as it has actually been. The use of modulated line radiation sources and synchronously tuned detection system, as proposed by Walsh, made the AAS technique highly specific and selective, but it obviously also made it a one-element-at-a-time technique, one of the most serious limitations of conventional AAS.
However, although commercial atomic absorption spectrometers have been built exclusively according to the principle proposed by Walsh for more that four decades, research on the use of continuum radiation sources. For AAS has continued throughout this period. The early publications in the field mainly took advantage of the instability and/or low energy output of hollow cathode lamps for a number of elements, or their unavailability for other elements, particularly the rare-earth elements, and demonstrated in this way the superiority of the continuum source approach. Some authors, however, even questioned the validity of Walsh’s approach, although the detection limits reported for those elements for which good line sources were available, were at least one order of magnitude inferior with a continuum source.
Onway
第8楼2010/03/10
In the following years, several groups investigated wavelength modulation, using AC scanning, oscillating interferometers or a combination of optical scanning and mechanical chopping in order to improve the signal-to-noise ratio (SNR) and the sensitivity of continuum source AAS (CS AAS). In the latter work, Elsner and Winefordner reported analytical curves that were linear over at least three orders of magnitude, and detection limits that were close to the theoretical values.
A kind of turning point in this early phase of CA AAS was the work of Keliher and Wohlers who for the first time used a high-resolution echelle grating spectrometer for CS AAS. The major limitation at that time was the 150W xenon lamp used as the continuum source, which had only a relatively low energy at wavelengths below 320nm, where most of the elements have their most sensitive lines. This work was then continued over the next 25 years by the groups of O’Haver and Harnly, who continuously improved the system, introducing wavelength modulation, a pulsed continuum source and a linear photodiode array detector. They also described the first, and up until now only, functional simultaneous multi-element atomic absorption spectrometer with a continuum source (SIMAAC), and showed the applicability of this system for a variety of practical analytical problems using flame and graphite furnace atomization. The only other ‘simultaneous’ CS AAS instruments describe in the literature used photodiode array detectors that covered a spectral range of 2.5nm and 10nm, respectively, and only elements that had absorption lines falling within this narrow spectral window could be detected simultaneously. This approach, obviously, cannot be considered a true simultaneous multi-element system.