

第1楼2005/03/30
Figures 9.1 and 9.2 show total trace metals and major contributing ions for cut parts and from PFA 1 resin pellets and extruded tubing following extraction.2
Fig 9.1: Total Trace Metals & Contributing Ions for Cut Parts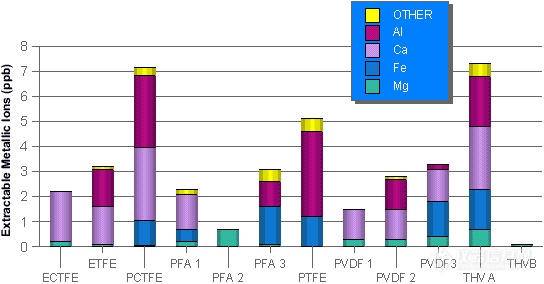
- Extracted for 5 days in 10% ultrapure HNO3 at 25°C -
Fig 9.2: Total Trace Metals & Major Contributing Ions from PFA 1 Resin Pellets and Tubing
- Extracted for 5 days in 2% ultrapure HNO3 at 25°C -
第3楼2005/03/30
High Purity Acids:
High purity acids have been commercially available for years with the major impurities typically less than 1 ppb. Distilling the acid yourself may offer some improvement in purity. If you are using more than 500 mL of mineral acid per month, you may want to consider the monetary savings of distilling the acid yourself. Diagrams 9.1 and 9.2 below show typical quartz and teflon stills.
Diagram 9.1: Pure Quartz Sub-Boiling Still
Diagram 9.2: All-Teflon Sub-Boiling Still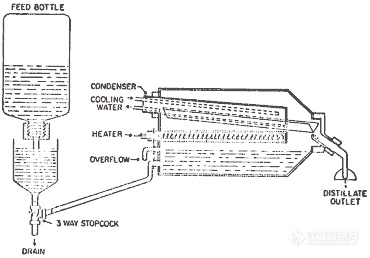
第8楼2005/03/30
Table 9.5: Impurity Concentration in Hydrofluoric Acid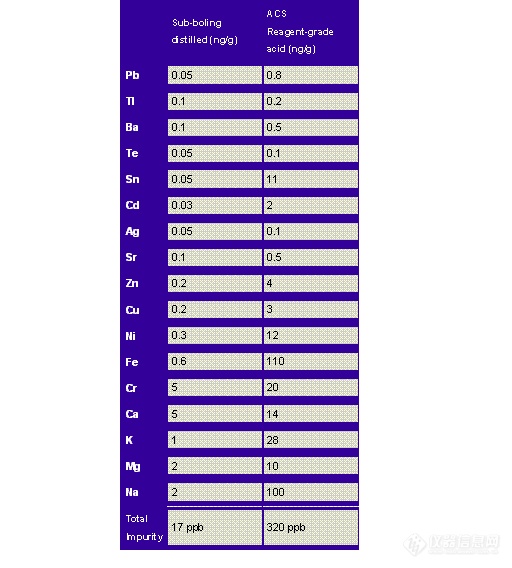
Other Reagents:
The "purity" situation for salts and other reagents is typically not as favorable as it is for water and acids. Typically, the highest purity solid that can be confirmed is 99.999% (5-9's or TMI 10ppm). This translates to 100 ppb total impurities for a 100 fold dilution (i.e. - 1 g of 5-9's pure reagent into 100 mL of solution). We recommend that you know your supplier's definition of "high purity". DO NOT use solid reagent grade materials when preparing samples for trace metals analysis whenever possible. If necessary, a blank should be performed to confirm the acceptability of reagents(s) or to identify problem elements / impurities. Purification of unacceptable reagents may be accomplished by mercury cathode electrolysis, extraction with dithiozone or cupferron, ion-exchange, or crystallization